The Translational Neural Repair and Regeneration Laboratory has access to the University of Georgia Bioimaging Research Center (BIRC) located in the Paul D. Coverdell Building for Biomedical and Health Sciences.
The BIRC is a new state-of-the-art 9,000 sq. ft. suite. A General Electric 32-channel MR750 3.0 Tesla MRI magnet permits multiple analysis techniques including structural MRI for studies involving tissue level changes, functional MRI (fMRI) for studies involving brain network activation in real time, magnetic resonance spectroscopy (MRS) for the study of chemical changes in the brain, and magnetic resonance angiography (MRA) for the study of vascular changes throughout the system. A 7 Tesla small animal magnet (Varian Medical Systems) is also available at the BIRC. This scanner has a 20 cm bore and is ideal for MRI of isolated porcine brains, poultry embryos, and in vivo rodent imaging. For more information on the BIRC please refer to https://birc.uga.edu/.
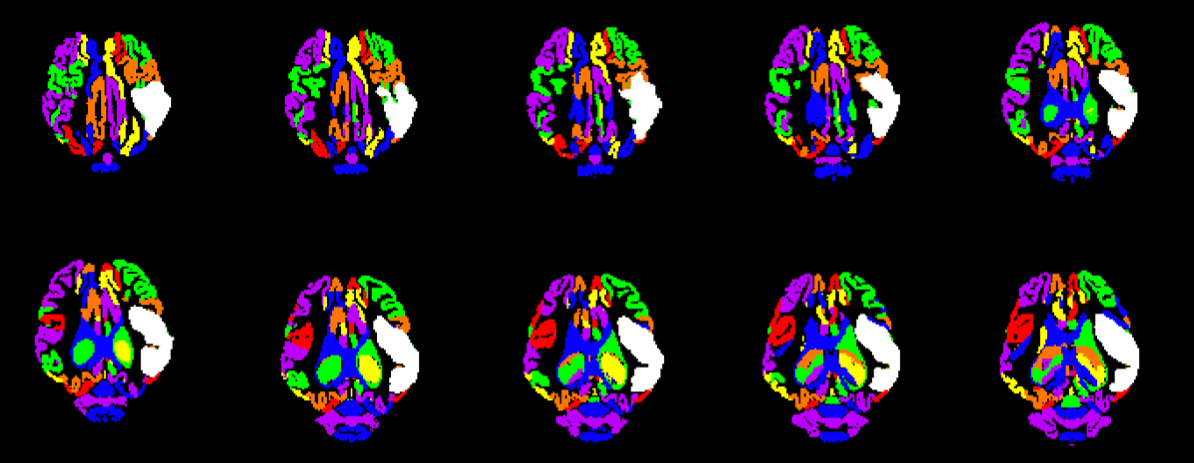
Structural MRI sequences have been specifically optimized by the TNRR Laboratory to evaluate tissue-level changes in real-time following neural injury and throughout recovery processes. Our protocols commonly include the following:
- High-resolution 3D T1-weighted (T1W), T2-weighted (T2W), T2 Fluid Attenuated Inversion Recovery (T2F), and T2Star (T2*) sequences to evaluate infarction, midline shift, tissue atrophy, parenchymal swelling, and intracerebral hemorrhage;
- Diffusion weighted imaging (DWI) and derived apparent diffusion coefficient (ADC) maps to measure edema, free extracellular and restricted interstitial water content, and ischemia, which often change significantly post-stroke and TBI due to cellular swelling;
- Fractional anisotropy (FA) to evaluate changes in white matter integrity as well as radial diffusivity (RD) for demyelination, mean diffusivity (MD) for white matter lesions, and diffusion tensor imaging (DTI) based tractography reconstructions for structural connectivity evaluations.
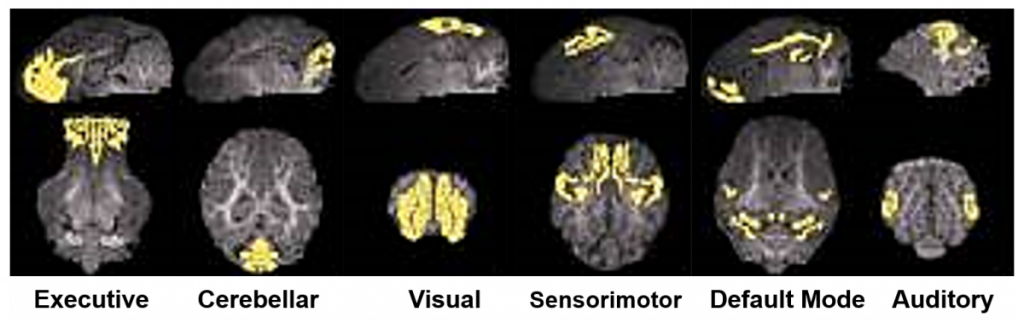
fMRI, or more specifically, resting state and task-based fMRI (rs-fMRI and tb-fMRI), are two highly sensitive, non-invasive imaging approaches that possess substantial clinical and preclinical utility as they can be used to assess functional connectivity following neural injury and in response to novel therapeutics. For example, the TNRR Laboratory has been able to characterize normal and TBI-perturbed resting state networks including the executive, cerebellar, visual, sensorimotor, default mode, and auditory networks1. The assessment of these networks in conjunction with cognitive, behavior, and motor function tests provide a comprehensive assessment of functional recovery following neural injury.
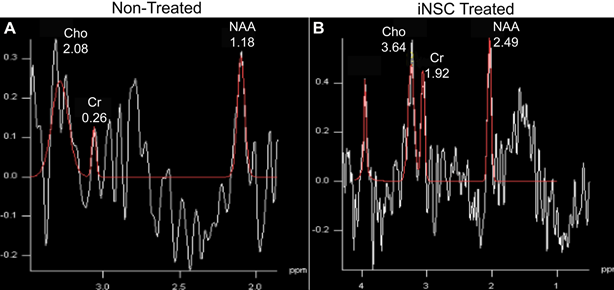
MRS enables the assessment of neurochemical changes in GABA (neurotransmitter), NAA (neuronal marker), creatine (Cr, a cell energy metabolite), and choline (Cho, marker of cellularity). For example, MRS assessment revealed an increase in select neurometabolites 24 hours post-stroke as well as 1, 4, and 12 weeks post-iNSC transplant2. Collectively, MRI and MRS enable the identification of key pathologies (e.g. cell death, microbleeds, loss of tissue integrity and connectivity) and provide metabolic biomarkers associated with neural injury and recovery.
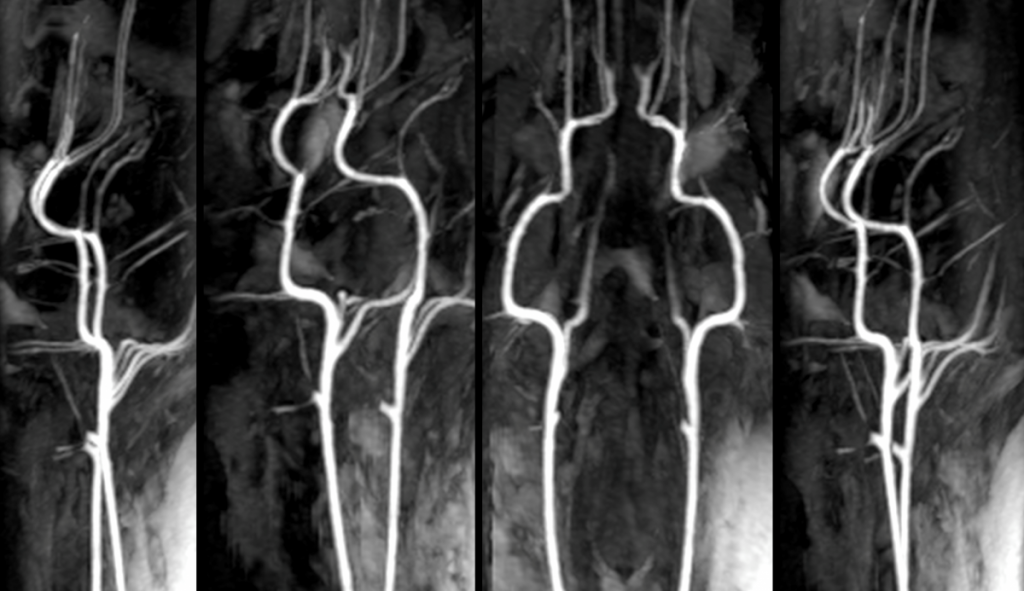
MRA is utilized to image blood vessels in order to evaluate the presence of stenosis, occlusions, aneurysms, or other abnormalities. In clinical applications, MRA-DWI mismatch helps to identify stroke patients who will likely benefit from reperfusion therapy3. The TNRR Laboratory has also utilized this sequence to pioneer a pig vascular cognitive impairment model.
The TNRR Laboratory analyzes all the above sequences in house utilizing advanced software platforms such as Osirix, 3D slicer, Matlab, MedInria, and FSL. These analyses help to increase the clinical relevance of the TNRR Laboratory’s porcine models as they are frequently utilized for comparable human analyses.
- 1Simchick, G., et al., Pig Brains Have Homologous Resting-State Networks with Human Brains. Brain Connect, 2019. 9(7): p. 566-579.
- 2Baker, E.W., et al., Induced Pluripotent Stem Cell-Derived Neural Stem Cell Therapy Enhances Recovery in an Ischemic Stroke Pig Model. Sci Rep, 2017. 7(1): p. 10075.
- 3Olivot, J. M., et al., The MRA-DWI mismatch identifies patients with stroke who are likely to benefit from reperfusion. Stroke, 2008. 39(9): p. 2491–2496.